the ROME - the Conference of The Regions has given the go-ahead to the appointment of Nicholas Rodriguez as the new director-general of the Agency of drug (Aifa). The Regions gave a favourable opinion to the proposal of the Minister of Health, Roberto Hope.
the Finish is a technical and very estimated that in the last few years, from 2014 onwards, has been the secretary of the Committee for the basic drugs of the Who. This is the body that writes the list of medications considered to be essential. The list contains the products, indeed indispensable, to the exclusion of the hundreds of duplicates, and also of medicines, unnecessary of which are sold, and often also refunded, in the health care systems of the developed Countries. Previously, he was responsible for the area of drugs of the Agency of healthcare services of Emilia-Romagna. In that role, he supported the fight of the Region against the pharmaceutical industry in the case Avasin-Lucentis, two drugs that work against macular degeneration, but only the second has the permission to that use because the company that produces the first (la Roche) has never request. Emilia decided to use the Avastin and had to surrender when the Aifa, at that time directed by Luca Pani, went sideways and stopped. Just Magrini was among those who worked on the appeal of the region against that decision. At the end of the line emiliana has prevailed, and the council of State and Antitrust have established that the two industries, in addition to Roche and Novartis, had agreed not to compete with each other. And on each other, called in their case by the Council of the State, it is also expressed by the Court of Justice of the european union in January 2018, which reaffirmed that "the agreement between the pharmaceutical groups Roche and Novartis, which aims to reduce the use in the field of ophthalmic medicine Avastin, and increase that of Lucentis could constitute a restriction of competition "by object"”.
He is among the founders of the Cochrane Italian, that is, the international organization that "collects and synthesizes the best scientific evidence to help you make informed decisions about therapeutic strategies". It is, therefore, a great sostenitotre of Ebm, evidence based medicine (medicine based on evidence) that is translated in the pharmaceutical field means that shall be used in medicinal products for which there is scientific evidence of effectiveness solid.
"we have Finally closed the issue of the appointment of the nominating committee for the Lea of the Agency for the medication, the Aifa and the higher health Institute - has explained the vice-president of the Conference of the Regions, Donato Toma , at the end of the work of the Conference, Then the conference of the Regions has made its designations in perfect unanimity".
the Finish is a technical and very estimated that in the last few years, from 2014 onwards, has been the secretary of the Committee for the basic drugs of the Who. This is the body that writes the list of medications considered to be essential. The list contains the products, indeed indispensable, to the exclusion of the hundreds of duplicates, and also of medicines, unnecessary of which are sold, and often also refunded, in the health care systems of the developed Countries. Previously, he was responsible for the area of drugs of the Agency of healthcare services of Emilia-Romagna. In that role, he supported the fight of the Region against the pharmaceutical industry in the case Avasin-Lucentis, two drugs that work against macular degeneration, but only the second has the permission to that use because the company that produces the first (la Roche) has never request. Emilia decided to use the Avastin and had to surrender when the Aifa, at that time directed by Luca Pani, went sideways and stopped. Just Magrini was among those who worked on the appeal of the region against that decision. At the end of the line emiliana has prevailed, and the council of State and Antitrust have established that the two industries, in addition to Roche and Novartis, had agreed not to compete with each other. And on each other, called in their case by the Council of the State, it is also expressed by the Court of Justice of the european union in January 2018, which reaffirmed that "the agreement between the pharmaceutical groups Roche and Novartis, which aims to reduce the use in the field of ophthalmic medicine Avastin, and increase that of Lucentis could constitute a restriction of competition "by object"”.
He is among the founders of the Cochrane Italian, that is, the international organization that "collects and synthesizes the best scientific evidence to help you make informed decisions about therapeutic strategies". It is, therefore, a great sostenitotre of Ebm, evidence based medicine (medicine based on evidence) that is translated in the pharmaceutical field means that shall be used in medicinal products for which there is scientific evidence of effectiveness solid.
"we have Finally closed the issue of the appointment of the nominating committee for the Lea of the Agency for the medication, the Aifa and the higher health Institute - has explained the vice-president of the Conference of the Regions, Donato Toma , at the end of the work of the Conference, Then the conference of the Regions has made its designations in perfect unanimity".
"The Republic will fight always in defense of the freedom of information, to its readers and to all those who have at heart the principles of democracy and civil coexistence"
Carlo Verdelli SUBSCRIBERS TO REPUBLIC © Reproduction reserved Today on Delrio: “On migrants, we do not changes soft. We need a new law” The habit of pretending nothing is Pd, is already an air of congress. Gori Bonaccini here who think primary, No, it's not the economy M5S, change at the top of Rousseau. And Of the Baptist there is a new rolethe Republic

 Gaza: under the spotlight, the Israeli-Palestinian conflict shakes up the Eurovision contest
Gaza: under the spotlight, the Israeli-Palestinian conflict shakes up the Eurovision contest Black soldier killed by a police officer in the United States: the sheriff publishes the video of the arrest
Black soldier killed by a police officer in the United States: the sheriff publishes the video of the arrest In Malmö, the Eurovision party transformed into entrenched camps
In Malmö, the Eurovision party transformed into entrenched camps In Russia, Vladimir Putin stigmatizes “Western elites”
In Russia, Vladimir Putin stigmatizes “Western elites” Fatal case of cholera in Mayotte: the epidemic is “contained”, assures the government
Fatal case of cholera in Mayotte: the epidemic is “contained”, assures the government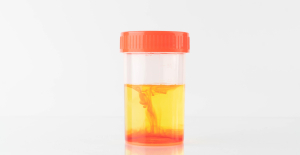 The presence of blood in the urine, a warning sign of bladder cancer
The presence of blood in the urine, a warning sign of bladder cancer A baby whose mother smoked during pregnancy will age more quickly
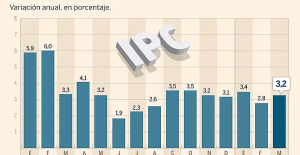
A baby whose mother smoked during pregnancy will age more quickly The euro zone economy grows in April at its best pace in almost a year but inflationary pressure increases
The euro zone economy grows in April at its best pace in almost a year but inflationary pressure increases Artificial intelligence lies, cheats and deceives us, and that's a problem, experts warn
Artificial intelligence lies, cheats and deceives us, and that's a problem, experts warn Google Cloud mistakenly deletes UniSuper fund account and deprives 600,000 Australians of their superannuation
Google Cloud mistakenly deletes UniSuper fund account and deprives 600,000 Australians of their superannuation IBM, Amazon, Hager... These record investments expected at the Choose France summit
IBM, Amazon, Hager... These record investments expected at the Choose France summit Boeing's black streak: a second Air France flight diverted in three days for “a smell of heat”
Boeing's black streak: a second Air France flight diverted in three days for “a smell of heat” “I want to apologize”: Kendji Girac speaks for the first time since his injury
“I want to apologize”: Kendji Girac speaks for the first time since his injury Gollum at the heart of the next Lord of the Rings film and controversies
Gollum at the heart of the next Lord of the Rings film and controversies In Paris, the redevelopment of the squares between Bastille and the Saint-Martin canal displeases some residents
In Paris, the redevelopment of the squares between Bastille and the Saint-Martin canal displeases some residents Cate Blanchett in majesty at the San Sebastian Festival
Cate Blanchett in majesty at the San Sebastian Festival Omoda 7, another Chinese car that could be manufactured in Spain
Omoda 7, another Chinese car that could be manufactured in Spain BYD chooses CA Auto Bank as financial partner in Spain
BYD chooses CA Auto Bank as financial partner in Spain Tesla and Baidu sign key agreement to boost development of autonomous driving
Tesla and Baidu sign key agreement to boost development of autonomous driving Skoda Kodiaq 2024: a 'beast' plug-in hybrid SUV
Skoda Kodiaq 2024: a 'beast' plug-in hybrid SUV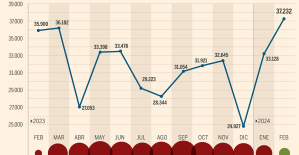 The home mortgage firm rises 3.8% in February and the average interest moderates to 3.33%
The home mortgage firm rises 3.8% in February and the average interest moderates to 3.33%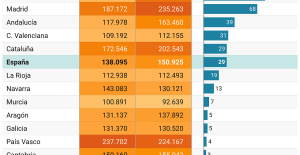 This is how housing prices have changed in Spain in the last decade
This is how housing prices have changed in Spain in the last decade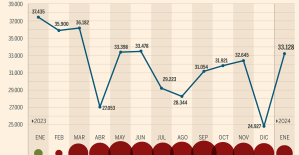 The home mortgage firm drops 10% in January and interest soars to 3.46%
The home mortgage firm drops 10% in January and interest soars to 3.46% The jewel of the Rocío de Nagüeles urbanization: a dream villa in Marbella
The jewel of the Rocío de Nagüeles urbanization: a dream villa in Marbella Diving into the secrets of the National Assembly
Diving into the secrets of the National Assembly Institutions: senators want to restore the accumulation of mandates and put an end to the automatic presence of ex-presidents on the Constitutional Council
Institutions: senators want to restore the accumulation of mandates and put an end to the automatic presence of ex-presidents on the Constitutional Council Europeans: David Lisnard expresses his “essential and vital” support for François-Xavier Bellamy
Europeans: David Lisnard expresses his “essential and vital” support for François-Xavier Bellamy Facing Jordan Bardella, the popularity match turns to Gabriel Attal’s advantage
Facing Jordan Bardella, the popularity match turns to Gabriel Attal’s advantage These French cities that will boycott the World Cup in Qatar
These French cities that will boycott the World Cup in Qatar Tennis: Djokovic knocked out by a gourd after his match in Rome (on video)
Tennis: Djokovic knocked out by a gourd after his match in Rome (on video) Handball: the Blues gently begin their Olympic preparation against the United States
Handball: the Blues gently begin their Olympic preparation against the United States WRC: Rovanperä leads the Rallye du Portugal ahead of Ogier
WRC: Rovanperä leads the Rallye du Portugal ahead of Ogier Ligue 2: Auxerre validates its return to Ligue 1 after a draw in Amiens
Ligue 2: Auxerre validates its return to Ligue 1 after a draw in Amiens