The vaccination campaign offered to middle school students in 5th grade against human papillomavirus (HPV), a virus causing many cancers such as cervical cancer, was launched at the beginning of October in France. But on October 19, the fatal fall of an adolescent following post-vaccination discomfort in a college pushed the medicines agency (ANSM) to recommend a new protocol: adolescents must now remain lying or sitting on the ground for the fifteen minutes following the injection.
A note from the ARS (Regional Health Agency) Île-de-France published in June explained that students had to be seated on “chairs” or “armchairs”, and recommended lying down students who were unwell. But in a document published on its website on Tuesday, the National Agency for the Safety of Medicines and Health Products (ANSM) updated the “post-vaccination surveillance” instructions for health professionals. As one of the main side effects of the vaccine is the risk of “discomfort”, “it is important that simple measures are put in place to avoid any injury: vaccinated people must remain lying down (on floor mats or blankets ) or sitting on the ground leaning against a wall in a clear space,” explains the agency. Until now, the instructions for vaccination in college recommended monitoring of students within a quarter of an hour following the injection, without specifying in what context.
This change of instructions comes following the death after a heavy fall of a 5th grade student from Saint-Dominique college in Saint-Herblain near Nantes (Loire-Atlantique) at the end of October, the ANSM confirmed to AFP on Tuesday. The Nantes public prosecutor's office, which opened an investigation into manslaughter, told AFP that “the schoolboy, who was not feeling well shortly after the injection, was seated on a chair and fell backwards. on the head after feeling unwell. The ANSM specifies that post-vaccination discomfort, sometimes brief losses of consciousness, are “uncommon and quickly resolved, may correspond to a psychogenic reaction to the injection” and “may be accompanied by tremors or stiffness”. The vaccine product itself is not the cause.

 Sabadell rejects the merger with BBVA and will fight to remain alone
Sabadell rejects the merger with BBVA and will fight to remain alone In Germany, the far left wants to cap the price of “doner kebabs”
In Germany, the far left wants to cap the price of “doner kebabs” Israel-Hamas war: Gaza between hope of truce and fear of Israeli offensive in the South
Israel-Hamas war: Gaza between hope of truce and fear of Israeli offensive in the South “Mom, Dad, please don’t die”: in the United States, a nine-year-old child saves the lives of his parents injured in a tornado
“Mom, Dad, please don’t die”: in the United States, a nine-year-old child saves the lives of his parents injured in a tornado The presence of blood in the urine, a warning sign of bladder cancer
The presence of blood in the urine, a warning sign of bladder cancer A baby whose mother smoked during pregnancy will age more quickly
A baby whose mother smoked during pregnancy will age more quickly The euro zone economy grows in April at its best pace in almost a year but inflationary pressure increases
The euro zone economy grows in April at its best pace in almost a year but inflationary pressure increases Children born thanks to PMA do not have more cancers than others
Children born thanks to PMA do not have more cancers than others “House of the Dragon”, “Succession”… Max, the new streaming platform from HBO and Discovery, launched in France on June 11
“House of the Dragon”, “Succession”… Max, the new streaming platform from HBO and Discovery, launched in France on June 11 The A13 motorway will finally reopen this Friday, in one direction only
The A13 motorway will finally reopen this Friday, in one direction only TNT commission of inquiry: tensions between LFI deputies and Macronists before the vote on the report
TNT commission of inquiry: tensions between LFI deputies and Macronists before the vote on the report Apple unveils a new, more efficient iPad range
Apple unveils a new, more efficient iPad range The Gaza War invites itself to the 2024 Pulitzer Prizes
The Gaza War invites itself to the 2024 Pulitzer Prizes Judith Godrèche presents a short film on sexual violence in Cannes
Judith Godrèche presents a short film on sexual violence in Cannes Kevin Spacey: new trial in sight in London for the American actor, for sexual assault
Kevin Spacey: new trial in sight in London for the American actor, for sexual assault Taylor Swift fans make London pub Black Dog their new place of pilgrimage
Taylor Swift fans make London pub Black Dog their new place of pilgrimage Omoda 7, another Chinese car that could be manufactured in Spain
Omoda 7, another Chinese car that could be manufactured in Spain BYD chooses CA Auto Bank as financial partner in Spain
BYD chooses CA Auto Bank as financial partner in Spain Tesla and Baidu sign key agreement to boost development of autonomous driving
Tesla and Baidu sign key agreement to boost development of autonomous driving Skoda Kodiaq 2024: a 'beast' plug-in hybrid SUV
Skoda Kodiaq 2024: a 'beast' plug-in hybrid SUV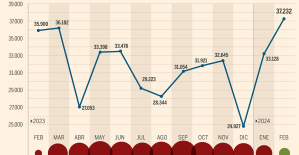 The home mortgage firm rises 3.8% in February and the average interest moderates to 3.33%
The home mortgage firm rises 3.8% in February and the average interest moderates to 3.33%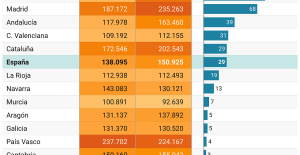 This is how housing prices have changed in Spain in the last decade
This is how housing prices have changed in Spain in the last decade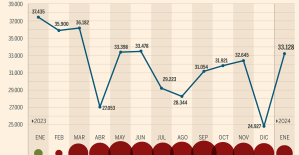 The home mortgage firm drops 10% in January and interest soars to 3.46%
The home mortgage firm drops 10% in January and interest soars to 3.46% The jewel of the Rocío de Nagüeles urbanization: a dream villa in Marbella
The jewel of the Rocío de Nagüeles urbanization: a dream villa in Marbella Institutions: senators want to restore the accumulation of mandates and put an end to the automatic presence of ex-presidents on the Constitutional Council
Institutions: senators want to restore the accumulation of mandates and put an end to the automatic presence of ex-presidents on the Constitutional Council Europeans: David Lisnard expresses his “essential and vital” support for François-Xavier Bellamy
Europeans: David Lisnard expresses his “essential and vital” support for François-Xavier Bellamy Facing Jordan Bardella, the popularity match turns to Gabriel Attal’s advantage
Facing Jordan Bardella, the popularity match turns to Gabriel Attal’s advantage Europeans: a senior official on the National Rally list
Europeans: a senior official on the National Rally list These French cities that will boycott the World Cup in Qatar
These French cities that will boycott the World Cup in Qatar “The future is for us”: “disappointed” and “proud” at the same time, Al-Khelaïfi sees the glass half full after the elimination of PSG
“The future is for us”: “disappointed” and “proud” at the same time, Al-Khelaïfi sees the glass half full after the elimination of PSG PSG: “Since January, these have not been my best matches,” agrees Zaire-Emery, who promises to “come back stronger”
PSG: “Since January, these have not been my best matches,” agrees Zaire-Emery, who promises to “come back stronger” “What is this question, honestly?” : Nasser Al-Khelaïfi (very) annoyed after PSG-Dortmund
“What is this question, honestly?” : Nasser Al-Khelaïfi (very) annoyed after PSG-Dortmund “I am the guy who has to score the goals”: Mbappé does not hide and assumes responsibility after PSG’s exit
“I am the guy who has to score the goals”: Mbappé does not hide and assumes responsibility after PSG’s exit