Reduce the number of high-risk pregnancies by better informing mothers and doctors: the National Agency for the Safety of Medicines and Health Products (ANSM) unveils on Wednesday November 29 a new overview of the risks linked to the consumption of antiepileptics.
“Four years after a first report, the panorama of antiepileptic drugs and the risks for the fetus has been modified by new data since 2019. New risks, previously suspected or unknown, are identified for drugs, which sometimes had other risks known,” explains Philippe Vella, medical director of the ANSM, to AFP.
The objective is to classify antiepileptic drugs according to the level of risk for the fetus, and to best inform prescribing doctors and patients. Valproate (Depakine and generics, Micropakine, Depakote and its generic Divalcote, Depamide) remains “the most at risk” if taken during pregnancy. This antiepileptic drug “leads to the most malformations in children, with a risk multiplied by 4 to 5 (11%) compared to the risk without treatment”, and “carries a high risk of neurodevelopmental disorders (30 to 40% of children exposed in utero),” detailed the agency.
And, from now on, its intake by the father, in the three months before conception, is also suspected of being a neurodevelopmental risk for the fetus, recalls the ANSM. While awaiting the conclusions of a European evaluation, it has already informed doctors and patients. One of the major changes made by the medicines agency concerns topiramate, sold under the brand Epitomax by the Janssen laboratory but also as a generic by other manufacturers and a source of growing concern for more than a year.
If the risk of major malformations (hare lip, poor placement of the urethra on the penis, very low birth weight, etc.) was "known" for this medication, the risk of neurodevelopmental disorders is "confirmed", according to the report. This risk is increased by two to three times compared to children born to epileptic mothers without treatment.
According to a study, carried out using the health data of several million Scandinavian women and published at the end of May 2022, the risk of intellectual disability is more than tripled in children whose mother took topiramate during pregnancy and the risk of disorders autism multiplied almost threefold.
In its wake, the ANSM asked its European counterpart, the European Medicines Agency (EMA), to reassess the conditions for prescribing topiramate and, among other things, called for this antiepileptic drug to be used only in pregnant women. “in case of absolute necessity”. Another change in the panorama drawn up by the ANSM: pregabalin (Lyrica and generics) presents a “confirmed” risk of major malformation in children - multiplied by almost 1.5 -, only “suspected” in 2019.
With carbamazepine (Tegretol and generics), whose risk of major malformation was “known” (multiplied by 2 to 3), there is also “a possible increase” in the risk of neurodevelopmental disorders. Of the twenty antiepileptic drugs reviewed, the risks of other treatments are not changed compared to 2019, at this stage of knowledge.
In recent years, the ANSM has tightened its policy towards antiepileptic drugs deemed to pose a risk during pregnancy. The involvement of several of these drugs in fetal disorders has been proven, notably for sodium valproate, or Depakine from Sanofi, at the heart of a scandal fueling several legal proceedings.
But in some women, only these treatments prove effective against epilepsy. It is therefore up to doctors alone to assess whether the risks represented by the attacks are high enough to compensate for those that the treatment poses to the unborn child.
The ANSM, however, provides patients with an information sheet summarizing the known risks of antiepileptic drugs for the future child and giving various advice, in particular on methods of contraception to adapt - certain antiepileptic drugs can reduce the effectiveness of the pill.

 Finding yourself face to face with a man or a bear? The debate that shakes up social networks
Finding yourself face to face with a man or a bear? The debate that shakes up social networks Sabadell rejects the merger with BBVA and will fight to remain alone
Sabadell rejects the merger with BBVA and will fight to remain alone In Germany, the far left wants to cap the price of “doner kebabs”
In Germany, the far left wants to cap the price of “doner kebabs” Israel-Hamas war: Gaza between hope of truce and fear of Israeli offensive in the South
Israel-Hamas war: Gaza between hope of truce and fear of Israeli offensive in the South The presence of blood in the urine, a warning sign of bladder cancer
The presence of blood in the urine, a warning sign of bladder cancer A baby whose mother smoked during pregnancy will age more quickly
A baby whose mother smoked during pregnancy will age more quickly The euro zone economy grows in April at its best pace in almost a year but inflationary pressure increases
The euro zone economy grows in April at its best pace in almost a year but inflationary pressure increases Children born thanks to PMA do not have more cancers than others
Children born thanks to PMA do not have more cancers than others “The prices are astronomical”: to enjoy the Olympics with family, Sabrina spent... 8,000 euros
“The prices are astronomical”: to enjoy the Olympics with family, Sabrina spent... 8,000 euros “House of the Dragon”, “Succession”… Max, the new streaming platform from HBO and Discovery, launched in France on June 11
“House of the Dragon”, “Succession”… Max, the new streaming platform from HBO and Discovery, launched in France on June 11 The A13 motorway will finally reopen this Friday, in one direction only
The A13 motorway will finally reopen this Friday, in one direction only TNT commission of inquiry: tensions between LFI deputies and Macronists before the vote on the report
TNT commission of inquiry: tensions between LFI deputies and Macronists before the vote on the report The Coubertin Spirit, Eternal Memory, Planet of the Apes... Films to watch this week
The Coubertin Spirit, Eternal Memory, Planet of the Apes... Films to watch this week Rappers Drake and Kendrick Lamar vie for the spotlight
Rappers Drake and Kendrick Lamar vie for the spotlight In Istanbul, the ancient Byzantine church of Saint-Sauveur-in-Chora reopens for Muslim worship
In Istanbul, the ancient Byzantine church of Saint-Sauveur-in-Chora reopens for Muslim worship David Castello-Lopes: a fresh wind blows on the one-man show with his show Authentique
David Castello-Lopes: a fresh wind blows on the one-man show with his show Authentique Omoda 7, another Chinese car that could be manufactured in Spain
Omoda 7, another Chinese car that could be manufactured in Spain BYD chooses CA Auto Bank as financial partner in Spain
BYD chooses CA Auto Bank as financial partner in Spain Tesla and Baidu sign key agreement to boost development of autonomous driving
Tesla and Baidu sign key agreement to boost development of autonomous driving Skoda Kodiaq 2024: a 'beast' plug-in hybrid SUV
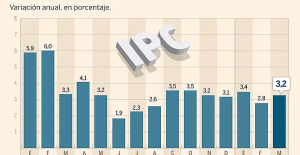
Skoda Kodiaq 2024: a 'beast' plug-in hybrid SUV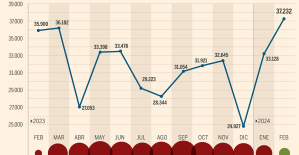 The home mortgage firm rises 3.8% in February and the average interest moderates to 3.33%
The home mortgage firm rises 3.8% in February and the average interest moderates to 3.33%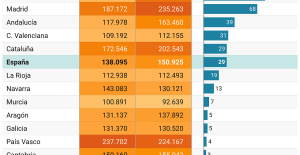 This is how housing prices have changed in Spain in the last decade
This is how housing prices have changed in Spain in the last decade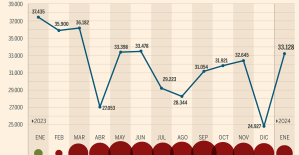 The home mortgage firm drops 10% in January and interest soars to 3.46%
The home mortgage firm drops 10% in January and interest soars to 3.46% The jewel of the Rocío de Nagüeles urbanization: a dream villa in Marbella
The jewel of the Rocío de Nagüeles urbanization: a dream villa in Marbella Institutions: senators want to restore the accumulation of mandates and put an end to the automatic presence of ex-presidents on the Constitutional Council
Institutions: senators want to restore the accumulation of mandates and put an end to the automatic presence of ex-presidents on the Constitutional Council Europeans: David Lisnard expresses his “essential and vital” support for François-Xavier Bellamy
Europeans: David Lisnard expresses his “essential and vital” support for François-Xavier Bellamy Facing Jordan Bardella, the popularity match turns to Gabriel Attal’s advantage
Facing Jordan Bardella, the popularity match turns to Gabriel Attal’s advantage Europeans: a senior official on the National Rally list
Europeans: a senior official on the National Rally list These French cities that will boycott the World Cup in Qatar
These French cities that will boycott the World Cup in Qatar Ligue 1: at what time and on which channel to watch the Multiplex on Sunday?
Ligue 1: at what time and on which channel to watch the Multiplex on Sunday? PSG-Dortmund: Hummels’ (big) tackle to “teams who wanted to play against” BVB
PSG-Dortmund: Hummels’ (big) tackle to “teams who wanted to play against” BVB Top 14: Pierre Bochaton extends with UBB until 2027
Top 14: Pierre Bochaton extends with UBB until 2027 Tennis: Monfils beaten, Atmane surprises and advances to the 2nd round in Rome
Tennis: Monfils beaten, Atmane surprises and advances to the 2nd round in Rome