Sandrine Sarrazin is an Inserm Research Officer at the Marseille-Luminy Immunology Center, and a guest researcher at the Center for Regenerative Therapies TU Dresden (Germany). Michael H. Sieweke is a research director at the CNRS and the Alexander-von-Humboldt Professor Center for Regenerative Therapies Technische Universität Dresden (Germany).
The immune system never stops revealing its secrets. In recent decades, fundamental research into understanding its complexity has regularly led to spectacular therapeutic advances, such as immunotherapy and more recently RNA vaccination. And over the past fifteen years, our work has also highlighted the unexpected and decisive role of blood stem cells, or “hematopoietic stem cells”, in the establishment of an effective and rapid immune response.
When we talk about immunity, many people do not automatically think of these cells. Nestled in the heart of the bone marrow, they are at the origin of all blood cells, multiplying and differentiating to produce red blood cells and platelets, but also white blood cells (lymphocytes, macrophages, neutrophils, etc.) , major players in the fight against infections. But it turned out that hematopoietic stem cells are much more than just a passive producer of blood cells...
Also read: Blood, a window open to our health
In 2009, while we were looking for something completely different, we became interested in blood stem cells. We wanted to understand how the maturation of macrophages, these “guardian” white blood cells of our organs, is controlled. We inactivated a gene that we thought was important for macrophages, but it was in blood stem cells that we discovered an important role!
We then understood what constituted the foundations of our work: stem cells do not always engage randomly in this or that differentiation pathway. Contrary to what we thought until now, this process can also take place under the direct influence of signals, both internal to the stem cells but also from the environment.
However, we did not know if in the event of an infection, blood stem cells were able to identify that it was an emergency situation and produce white blood cells on demand capable of fighting against pathogens. It took four years to show that a molecule called M-CSF, produced in large quantities by the body during an infection, is recognized by blood stem cells. Faced with this signal, they rush to quickly produce the white blood cells best suited to the situation. Better yet, blood stem cells are capable of recognizing certain pathogens directly thanks to specific receptors on their surface.
Also read: Stem cells: an old promise... which is coming true with very small steps
Finally, we must also mention what is perhaps the study that has moved the most forward: in 2020, we showed in animal models that blood stem cells, like other immune cells , possess a form of “memory” that we did not suspect until now: when they are exposed for the first time to a microbe, they are better equipped to face a future infection, by producing the necessary white blood cells even more quickly. And this memory lasts long term!
All of these discoveries clearly open the way to a change in status for the blood stem cell, making it go from queen of a deaf and blind hive to what is happening outside, to that of a true sentinel capable to detect an emergency situation and produce the workers best suited to respond to it.
But these advances, major on a fundamental level, could not have a real health and societal impact if they were not also accompanied by concrete therapeutic advances. And there are short-term prospects: it is in particular on the M-CSF molecule, the importance of which we have shown, that we have many hopes.
This molecule has already been tested in the United States and used as a drug in Japan in the 1990s for the treatment, for example, of certain cancers or neutrophil deficiency, a subtype of white blood cells. Unfortunately, due to manufacturing difficulties, its use was abandoned. Since then, the method of manufacturing M-CSF has been improved and we believe that it could have a much broader therapeutic interest than initially imagined, particularly for certain patients whose immunity is weakened.
Our work has so far focused on models of bone marrow transplants indicated in the treatment of certain blood cancers, pathologies of the bone marrow or the immune system, which affect more than 2,000 people in France every year. While these transplants save lives, they are also accompanied by complications due to the weakening of the patient's immune system and therefore increased exposure to attacks by pathogens. We have shown that in transplanted mice, injections of M-CSF can stimulate the production of white blood cells with a protective effect against infections. The effect is much greater than that of another molecule called G-CSF, already used clinically as an anti-infective agent. Indeed, M-CSF is capable of stimulating the differentiation of blood stem cells much further upstream, which has the advantage of producing, immediately after the transplant, a greater number of macrophages and other white blood cells, programmed to attack possible pathogens. Work is now continuing to establish the clinical usefulness of M-CSF in transplant patients or in other pathologies where the immune system is weakened.
Blood stem cells are now recognized as a central element and key regulator of the immune response. By exploiting their “immune memory” capacity, we could put these cells at the heart of new vaccine approaches. In the short term, their clinical application could be amplified by the use of M-CSF, an underestimated molecule that could find a second life as a drug.
*The authors declare that they have no interest in the subject matter.

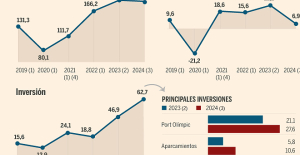 B:SM will break its investment record this year with 62 million euros
B:SM will break its investment record this year with 62 million euros War in Ukraine: when kyiv attacks Russia with inflatable balloons loaded with explosives
War in Ukraine: when kyiv attacks Russia with inflatable balloons loaded with explosives United States: divided on the question of presidential immunity, the Supreme Court offers respite to Trump
United States: divided on the question of presidential immunity, the Supreme Court offers respite to Trump Maurizio Molinari: “the Scurati affair, a European injury”
Maurizio Molinari: “the Scurati affair, a European injury” First three cases of “native” cholera confirmed in Mayotte
First three cases of “native” cholera confirmed in Mayotte Meningitis: compulsory vaccination for babies will be extended in 2025
Meningitis: compulsory vaccination for babies will be extended in 2025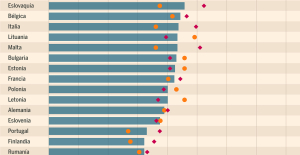 Spain is the country in the European Union with the most overqualified workers for their jobs
Spain is the country in the European Union with the most overqualified workers for their jobs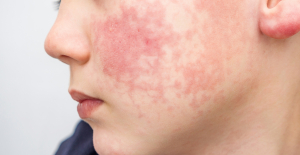 Parvovirus alert, the “fifth disease” of children which has already caused the death of five babies in 2024
Parvovirus alert, the “fifth disease” of children which has already caused the death of five babies in 2024 The A13 motorway will not reopen on May 1
The A13 motorway will not reopen on May 1 More than 1,500 items for less than 1 euro: the Dutch discounter Action opens a third store in Paris
More than 1,500 items for less than 1 euro: the Dutch discounter Action opens a third store in Paris 100 million euros in loans, water storage, Ecophyto plan… New measures from the executive towards farmers
100 million euros in loans, water storage, Ecophyto plan… New measures from the executive towards farmers “He is greatly responsible”: Philippe Martinez accuses Emmanuel Macron of having raised the RN
“He is greatly responsible”: Philippe Martinez accuses Emmanuel Macron of having raised the RN Les Galons de la BD dedicates War Photographers, a virtuoso album on the Spanish War
Les Galons de la BD dedicates War Photographers, a virtuoso album on the Spanish War Theater: Kevin, or the example of an academic failure
Theater: Kevin, or the example of an academic failure The eye of the INA: Jean Carmet, the thirst for life of a great actor
The eye of the INA: Jean Carmet, the thirst for life of a great actor The Nuc plus ultra: St Vincent the Texane and Neil Young the return
The Nuc plus ultra: St Vincent the Texane and Neil Young the return Skoda Kodiaq 2024: a 'beast' plug-in hybrid SUV
Skoda Kodiaq 2024: a 'beast' plug-in hybrid SUV Tesla launches a new Model Y with 600 km of autonomy at a "more accessible price"
Tesla launches a new Model Y with 600 km of autonomy at a "more accessible price" The 10 best-selling cars in March 2024 in Spain: sales fall due to Easter
The 10 best-selling cars in March 2024 in Spain: sales fall due to Easter A private jet company buys more than 100 flying cars
A private jet company buys more than 100 flying cars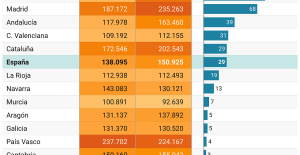 This is how housing prices have changed in Spain in the last decade
This is how housing prices have changed in Spain in the last decade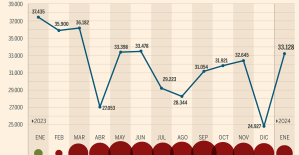 The home mortgage firm drops 10% in January and interest soars to 3.46%
The home mortgage firm drops 10% in January and interest soars to 3.46% The jewel of the Rocío de Nagüeles urbanization: a dream villa in Marbella
The jewel of the Rocío de Nagüeles urbanization: a dream villa in Marbella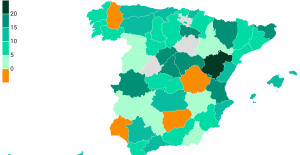 Rental prices grow by 7.3% in February: where does it go up and where does it go down?
Rental prices grow by 7.3% in February: where does it go up and where does it go down? Even on a mission for NATO, the Charles-de-Gaulle remains under French control, Lecornu responds to Mélenchon
Even on a mission for NATO, the Charles-de-Gaulle remains under French control, Lecornu responds to Mélenchon “Deadly Europe”, “economic decline”, immigration… What to remember from Emmanuel Macron’s speech at the Sorbonne
“Deadly Europe”, “economic decline”, immigration… What to remember from Emmanuel Macron’s speech at the Sorbonne Sale of Biogaran: The Republicans write to Emmanuel Macron
Sale of Biogaran: The Republicans write to Emmanuel Macron Europeans: “All those who claim that we don’t need Europe are liars”, criticizes Bayrou
Europeans: “All those who claim that we don’t need Europe are liars”, criticizes Bayrou These French cities that will boycott the World Cup in Qatar
These French cities that will boycott the World Cup in Qatar Paris 2024 Olympic Games: “It’s up to us to continue to honor what the Games are,” announces Estanguet
Paris 2024 Olympic Games: “It’s up to us to continue to honor what the Games are,” announces Estanguet MotoGP: Marc Marquez takes pole position in Spain
MotoGP: Marc Marquez takes pole position in Spain Ligue 1: Brest wants to play the European Cup at the Stade Francis-Le Blé
Ligue 1: Brest wants to play the European Cup at the Stade Francis-Le Blé Tennis: Tsitsipas released as soon as he entered the competition in Madrid
Tennis: Tsitsipas released as soon as he entered the competition in Madrid