for the First time a patient has been treated with a drug, the researchers specially for them have developed. A girl from the USA, Mila, suffers from a rare genetic disorder that leaves your nerves in the brain die off. Since February of 2018, Doctors inject a substance that consists of specially-produced molecules, so-called Antisense oligonucleotides (ASO). These new agents allow for the first time, to treat rare genetic disorders. The Problem is that For every genetic defect of its own ASO need to be made, such as in a colorful carpet holes with a cotton thread to mend can be, but depending on the pattern of a different color is needed.
The question is: Who pays for this kind of personalized medicine ? Pharmaceutical companies are hardly interested, if only a handful of patients suffering from the same genetic defect, and also the health insurance companies not to Finance such projects. The development can easily cost well over a Million Swiss francs per case.
time is of The essence
so What can parents do if the Doctors tell you that there is no therapy for your child? Some collect the money and ask researchers for help. Another possibility you have not currently. It was in the case of Mila the case in the USA, and also in Switzerland, the parents of Valeria have brought hundreds of thousands of francs for the therapy of your daughter. The toddler has a rare genetic disorder that leads to the fact that you can't develop mentally and motor-normal.
It may not be the case that parents themselves must take the drugs-development for your own child in the Hand.
The Dilemma in the development of personalized agents is, on the one hand, time is of the essence, because of the evil diseases move this fast (like Mila), or because, as early as possible therapies promise of success, are (as Valeria). On the other hand, the substances have to be tested well before they are administered to patients. This includes Tests on cells and animals as well as the assessment by an approval authority. Already, there are accelerated approval processes for these experimental drugs for seriously ill patients. It's A Start. Is not regulated, for whom these drugs are manufactured.
Faster diagnosis, more favorable preparations
It may not be the case that parents themselves must take the medication development for your own child in the Hand. Because so rare, rare diseases are not. Although they concern by Definition not more than five patients per 10'000 people. Taking together, but all the victims who suffer from the up to 8000 known rare diseases, so 30 to 40 million are in Europe, in Switzerland, 580'000.
it is Important that rare diseases will be faster diagnosed. For Doctors and genetic Tests carried out trained. Mila got her diagnosis late, as she had already lost a lot of skills. In addition, researchers, drug manufacturers and authorities need to seek solutions, such as new drugs cheaper can be developed.
One possibility would be, instead of individual safety tests for each specific ASO, to allow standardized Tests for the entire class of substances. This would reduce the cost of the therapy verb, and faster to make. So that more children could benefit with rare diseases, and not only individual, whose parents collect money for it. These parents have the path also in the future Affected person preparing.
Created: 05.11.2019, 21:54 PM

 Germany: Man armed with machete enters university library and threatens staff
Germany: Man armed with machete enters university library and threatens staff His body naturally produces alcohol, he is acquitted after a drunk driving conviction
His body naturally produces alcohol, he is acquitted after a drunk driving conviction Who is David Pecker, the first key witness in Donald Trump's trial?
Who is David Pecker, the first key witness in Donald Trump's trial? What does the law on the expulsion of migrants to Rwanda adopted by the British Parliament contain?
What does the law on the expulsion of migrants to Rwanda adopted by the British Parliament contain?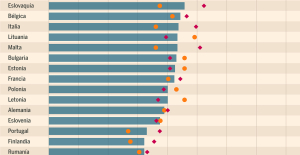 Spain is the country in the European Union with the most overqualified workers for their jobs
Spain is the country in the European Union with the most overqualified workers for their jobs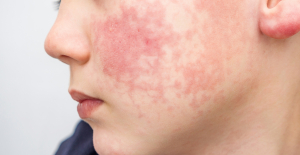 Parvovirus alert, the “fifth disease” of children which has already caused the death of five babies in 2024
Parvovirus alert, the “fifth disease” of children which has already caused the death of five babies in 2024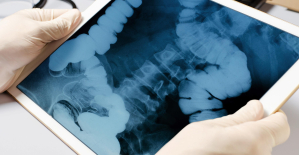 Colorectal cancer: what to watch out for in those under 50
Colorectal cancer: what to watch out for in those under 50 H5N1 virus: traces detected in pasteurized milk in the United States
H5N1 virus: traces detected in pasteurized milk in the United States Insurance: SFAM, subsidiary of Indexia, placed in compulsory liquidation
Insurance: SFAM, subsidiary of Indexia, placed in compulsory liquidation Under pressure from Brussels, TikTok deactivates the controversial mechanisms of its TikTok Lite application
Under pressure from Brussels, TikTok deactivates the controversial mechanisms of its TikTok Lite application “I can’t help but panic”: these passengers worried about incidents on Boeing
“I can’t help but panic”: these passengers worried about incidents on Boeing “I’m interested in knowing where the money that the State takes from me goes”: Bruno Le Maire’s strange pay slip sparks controversy
“I’m interested in knowing where the money that the State takes from me goes”: Bruno Le Maire’s strange pay slip sparks controversy 25 years later, the actors of Blair Witch Project are still demanding money to match the film's record profits
25 years later, the actors of Blair Witch Project are still demanding money to match the film's record profits At La Scala, Mathilde Charbonneaux is Madame M., Jacqueline Maillan
At La Scala, Mathilde Charbonneaux is Madame M., Jacqueline Maillan Deprived of Hollywood and Western music, Russia gives in to the charms of K-pop and manga
Deprived of Hollywood and Western music, Russia gives in to the charms of K-pop and manga Exhibition: Toni Grand, the incredible odyssey of a sculptural thinker
Exhibition: Toni Grand, the incredible odyssey of a sculptural thinker Skoda Kodiaq 2024: a 'beast' plug-in hybrid SUV
Skoda Kodiaq 2024: a 'beast' plug-in hybrid SUV Tesla launches a new Model Y with 600 km of autonomy at a "more accessible price"
Tesla launches a new Model Y with 600 km of autonomy at a "more accessible price" The 10 best-selling cars in March 2024 in Spain: sales fall due to Easter
The 10 best-selling cars in March 2024 in Spain: sales fall due to Easter A private jet company buys more than 100 flying cars
A private jet company buys more than 100 flying cars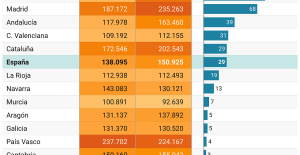 This is how housing prices have changed in Spain in the last decade
This is how housing prices have changed in Spain in the last decade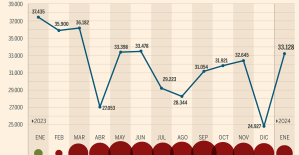 The home mortgage firm drops 10% in January and interest soars to 3.46%
The home mortgage firm drops 10% in January and interest soars to 3.46% The jewel of the Rocío de Nagüeles urbanization: a dream villa in Marbella
The jewel of the Rocío de Nagüeles urbanization: a dream villa in Marbella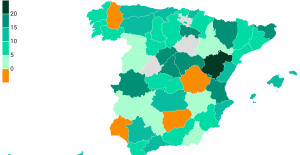 Rental prices grow by 7.3% in February: where does it go up and where does it go down?
Rental prices grow by 7.3% in February: where does it go up and where does it go down? Sale of Biogaran: The Republicans write to Emmanuel Macron
Sale of Biogaran: The Republicans write to Emmanuel Macron Europeans: “All those who claim that we don’t need Europe are liars”, criticizes Bayrou
Europeans: “All those who claim that we don’t need Europe are liars”, criticizes Bayrou With the promise of a “real burst of authority”, Gabriel Attal provokes the ire of the opposition
With the promise of a “real burst of authority”, Gabriel Attal provokes the ire of the opposition Europeans: the schedule of debates to follow between now and June 9
Europeans: the schedule of debates to follow between now and June 9 These French cities that will boycott the World Cup in Qatar
These French cities that will boycott the World Cup in Qatar Hand: Montpellier crushes Kiel and continues to dream of the Champions League
Hand: Montpellier crushes Kiel and continues to dream of the Champions League OM-Nice: a spectacular derby, Niçois timid despite their numerical superiority...The tops and the flops
OM-Nice: a spectacular derby, Niçois timid despite their numerical superiority...The tops and the flops Tennis: 1000 matches and 10 notable encounters by Richard Gasquet
Tennis: 1000 matches and 10 notable encounters by Richard Gasquet Tennis: first victory of the season on clay for Osaka in Madrid
Tennis: first victory of the season on clay for Osaka in Madrid


















