BY congress on gastrointestinal cancer of the American Society of Clinical Oncology (ASCO GI) in San Francisco come new data on the progress of the oncology precision. For patients who have a particular mutation - the gene fusion TRK - targeted drug larotrectinib showed a response rate of 43% in general, rising to 50% for the subgroup with colon cancer. In this case, all patients except one showed, as expected, also another feature of the genome: the high level of instability microsatellitare.
tumors with fusion TRK mergers TRK are of the chromosomal anomalies are rare and occur when one of the genes NTRK (receptor tyrosine kinase neurotrofica) is fused with another gene, giving rise to the production of abnormal protein. These fusion proteins TRK promote proliferation and cell survival, resulting in tumors with a fusion TRK (29 different types) that may form in many parts of the body. Larotrectinib is the first drug in the class of the inhibitors oral of TRK approved in Europe last September for all the cancers that have the merger, and without a specific indication of the organ.
Approved the first cancer drug with no indication of organ
The new data, The new analysis has regarded in all 14 patients with the mutation, of which 8 with colon cancer and the other 6 with cholangiocarcinoma, cancer of the pancreas, appendix and liver. In general, the progression-free survival median, after 19 months of follow-up was 5.3 months and the median time to response of 1.9 months.
“The responses we have observed with larotrectinib in gastrointestinal cancers are in line with the strong efficacy seen in lots of types different of cancer,” explains Salvatore Siena, full Professor of Medical Oncology at the University of Milan and Director of Oncology, Niguarda Cancer Center, Milano: “More than half of these patients had a tumor of the colon, most with high instability microsatellitare, further confirming that the fusion gene NTRK are more common in this subgroup. These data and the powerful responses that we observe with larotrectinib should encourage a wider application of genomic tests to identify gene mutations, especially in patients with colon cancer with high instability microsatellitare”.
Cancer, drugs without indication of the body: how to find patients right?
Larotrectinib is already approved in various Countries, including those of the European Union, for patients with solid tumors that have a fusion gene NTRK, metastatic, or in which surgical resection is likely to cause a severe morbidity and that have no satisfactory alternative treatment or in progression after therapy. This indication is approved under accelerated procedure on the basis of overall response rate and duration of response.
tumors with fusion TRK mergers TRK are of the chromosomal anomalies are rare and occur when one of the genes NTRK (receptor tyrosine kinase neurotrofica) is fused with another gene, giving rise to the production of abnormal protein. These fusion proteins TRK promote proliferation and cell survival, resulting in tumors with a fusion TRK (29 different types) that may form in many parts of the body. Larotrectinib is the first drug in the class of the inhibitors oral of TRK approved in Europe last September for all the cancers that have the merger, and without a specific indication of the organ.
Approved the first cancer drug with no indication of organ
The new data, The new analysis has regarded in all 14 patients with the mutation, of which 8 with colon cancer and the other 6 with cholangiocarcinoma, cancer of the pancreas, appendix and liver. In general, the progression-free survival median, after 19 months of follow-up was 5.3 months and the median time to response of 1.9 months.
“The responses we have observed with larotrectinib in gastrointestinal cancers are in line with the strong efficacy seen in lots of types different of cancer,” explains Salvatore Siena, full Professor of Medical Oncology at the University of Milan and Director of Oncology, Niguarda Cancer Center, Milano: “More than half of these patients had a tumor of the colon, most with high instability microsatellitare, further confirming that the fusion gene NTRK are more common in this subgroup. These data and the powerful responses that we observe with larotrectinib should encourage a wider application of genomic tests to identify gene mutations, especially in patients with colon cancer with high instability microsatellitare”.
Cancer, drugs without indication of the body: how to find patients right?
Larotrectinib is already approved in various Countries, including those of the European Union, for patients with solid tumors that have a fusion gene NTRK, metastatic, or in which surgical resection is likely to cause a severe morbidity and that have no satisfactory alternative treatment or in progression after therapy. This indication is approved under accelerated procedure on the basis of overall response rate and duration of response.
"The Republic will fight always in defense of the freedom of information, to its readers and to all those who have at heart the principles of democracy and civil coexistence"
Carlo Verdelli SUBSCRIBERS TO REPUBLIC © Reproduction reserved Today By the League and allies critical to Salvini. Giorgetti: errors in the countryside Elly Schlein: “Courage on migrants. Just follow the centre” Pd, Zingaretti says yes to Prodi: “I Want a party that is more open,” The majority without justice Bonafede delegation leader, but alliances split the 5Sthe Republic

 Germany: Man armed with machete enters university library and threatens staff
Germany: Man armed with machete enters university library and threatens staff His body naturally produces alcohol, he is acquitted after a drunk driving conviction
His body naturally produces alcohol, he is acquitted after a drunk driving conviction Who is David Pecker, the first key witness in Donald Trump's trial?
Who is David Pecker, the first key witness in Donald Trump's trial? What does the law on the expulsion of migrants to Rwanda adopted by the British Parliament contain?
What does the law on the expulsion of migrants to Rwanda adopted by the British Parliament contain?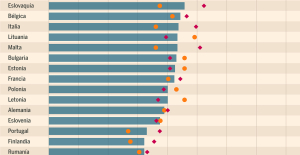 Spain is the country in the European Union with the most overqualified workers for their jobs
Spain is the country in the European Union with the most overqualified workers for their jobs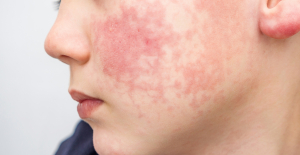 Parvovirus alert, the “fifth disease” of children which has already caused the death of five babies in 2024
Parvovirus alert, the “fifth disease” of children which has already caused the death of five babies in 2024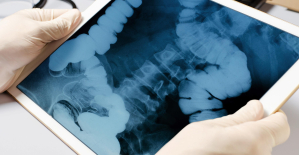 Colorectal cancer: what to watch out for in those under 50
Colorectal cancer: what to watch out for in those under 50 H5N1 virus: traces detected in pasteurized milk in the United States
H5N1 virus: traces detected in pasteurized milk in the United States Insurance: SFAM, subsidiary of Indexia, placed in compulsory liquidation
Insurance: SFAM, subsidiary of Indexia, placed in compulsory liquidation Under pressure from Brussels, TikTok deactivates the controversial mechanisms of its TikTok Lite application
Under pressure from Brussels, TikTok deactivates the controversial mechanisms of its TikTok Lite application “I can’t help but panic”: these passengers worried about incidents on Boeing
“I can’t help but panic”: these passengers worried about incidents on Boeing “I’m interested in knowing where the money that the State takes from me goes”: Bruno Le Maire’s strange pay slip sparks controversy
“I’m interested in knowing where the money that the State takes from me goes”: Bruno Le Maire’s strange pay slip sparks controversy 25 years later, the actors of Blair Witch Project are still demanding money to match the film's record profits
25 years later, the actors of Blair Witch Project are still demanding money to match the film's record profits At La Scala, Mathilde Charbonneaux is Madame M., Jacqueline Maillan
At La Scala, Mathilde Charbonneaux is Madame M., Jacqueline Maillan Deprived of Hollywood and Western music, Russia gives in to the charms of K-pop and manga
Deprived of Hollywood and Western music, Russia gives in to the charms of K-pop and manga Exhibition: Toni Grand, the incredible odyssey of a sculptural thinker
Exhibition: Toni Grand, the incredible odyssey of a sculptural thinker Skoda Kodiaq 2024: a 'beast' plug-in hybrid SUV
Skoda Kodiaq 2024: a 'beast' plug-in hybrid SUV Tesla launches a new Model Y with 600 km of autonomy at a "more accessible price"
Tesla launches a new Model Y with 600 km of autonomy at a "more accessible price" The 10 best-selling cars in March 2024 in Spain: sales fall due to Easter
The 10 best-selling cars in March 2024 in Spain: sales fall due to Easter A private jet company buys more than 100 flying cars
A private jet company buys more than 100 flying cars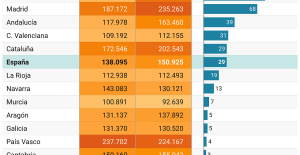 This is how housing prices have changed in Spain in the last decade
This is how housing prices have changed in Spain in the last decade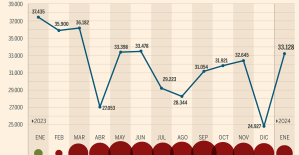 The home mortgage firm drops 10% in January and interest soars to 3.46%
The home mortgage firm drops 10% in January and interest soars to 3.46% The jewel of the Rocío de Nagüeles urbanization: a dream villa in Marbella
The jewel of the Rocío de Nagüeles urbanization: a dream villa in Marbella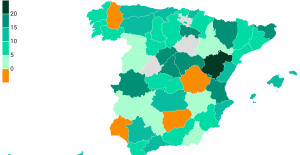 Rental prices grow by 7.3% in February: where does it go up and where does it go down?
Rental prices grow by 7.3% in February: where does it go up and where does it go down? Sale of Biogaran: The Republicans write to Emmanuel Macron
Sale of Biogaran: The Republicans write to Emmanuel Macron Europeans: “All those who claim that we don’t need Europe are liars”, criticizes Bayrou
Europeans: “All those who claim that we don’t need Europe are liars”, criticizes Bayrou With the promise of a “real burst of authority”, Gabriel Attal provokes the ire of the opposition
With the promise of a “real burst of authority”, Gabriel Attal provokes the ire of the opposition Europeans: the schedule of debates to follow between now and June 9
Europeans: the schedule of debates to follow between now and June 9 These French cities that will boycott the World Cup in Qatar
These French cities that will boycott the World Cup in Qatar Hand: Montpellier crushes Kiel and continues to dream of the Champions League
Hand: Montpellier crushes Kiel and continues to dream of the Champions League OM-Nice: a spectacular derby, Niçois timid despite their numerical superiority...The tops and the flops
OM-Nice: a spectacular derby, Niçois timid despite their numerical superiority...The tops and the flops Tennis: 1000 matches and 10 notable encounters by Richard Gasquet
Tennis: 1000 matches and 10 notable encounters by Richard Gasquet Tennis: first victory of the season on clay for Osaka in Madrid
Tennis: first victory of the season on clay for Osaka in Madrid


















