This meeting was held days after another advisory group, this one representing the Food and Drug Administration, had rejected a White House proposal to distribute third shots to almost everyone. The panel recommended booster doses only for seniors and people at high risk of contracting the virus.
The COVID-19 vaccines offer strong protection against serious illness, hospitalization, and death. However, the immunity to milder infections seems to be declining months after vaccination.
The FDA advisory panel's decision last week was just the beginning of the government's booster policy. The FDA must decide if it accepts the recommendation of its advisors and will allow Pfizer boosters.
If it does, then the CDC must make recommendations to whomever should receive the additional shots, after listening to the Advisory Committee on Immunization practices, whose meeting was set for Thursday.
Priority still remains to vaccinate unvaccinated people, who, according to the CDC, account for the majority of COVID-19-related cases. This is a situation that has been unprecedented since last winter. Nearly 55% of Americans are fully vaccinated. This is 182 million Americans.
"I want you to know that COVID-19 deaths in the United States in September 2021 are largely preventable with the primary series vaccines," Dr. Matthew Daley, a Kaiser Permanente Colorado researcher and CDC adviser, said Wednesday.
The meeting's main focus was on who should be considered high enough at risk to receive an additional dose. For example, should health care workers who have been exposed to the virus for a long time be eligible for boosters?
Another question was when boosters should be administered. Scientists debate whether boosters should be given after six or eight months.
Experts are divided about whether boosters are necessary because they believe the COVID-19 vaccines work as expected. It is not unusual for the body's virus-fighting antibodies (antivirals) to diminish over several months after vaccination. The body still has backup defenses against the virus.
The government is evaluating whether the prevention of "breakthrough" infection in fully vaccinated children could reduce virus transmission, protect young children not yet eligible for vaccination, and lessen the burden on overloaded health care systems.
The government will determine at a later time whether additional doses of Moderna and Johnson & Johnson vaccines are allowed.

 After 13 years of mission and seven successive leaders, the UN at an impasse in Libya
After 13 years of mission and seven successive leaders, the UN at an impasse in Libya Germany: search of AfD headquarters in Lower Saxony, amid accusations of embezzlement
Germany: search of AfD headquarters in Lower Saxony, amid accusations of embezzlement Faced with Iran, Israel plays appeasement and continues its shadow war
Faced with Iran, Israel plays appeasement and continues its shadow war Iran-Israel conflict: what we know about the events of the night after the explosions in Isfahan
Iran-Israel conflict: what we know about the events of the night after the explosions in Isfahan Sánchez condemns Iran's attack on Israel and calls for "containment" to avoid an escalation
Sánchez condemns Iran's attack on Israel and calls for "containment" to avoid an escalation China's GDP grows 5.3% in the first quarter, more than expected
China's GDP grows 5.3% in the first quarter, more than expected Alert on the return of whooping cough, a dangerous respiratory infection for babies
Alert on the return of whooping cough, a dangerous respiratory infection for babies Can relaxation, sophrology and meditation help with insomnia?
Can relaxation, sophrology and meditation help with insomnia? Vacation departures and returns: with the first crossovers, heavy traffic is expected this weekend
Vacation departures and returns: with the first crossovers, heavy traffic is expected this weekend “Têtu”, “Ideat”, “The Good Life”… The magazines of the I/O Media group resold to several buyers
“Têtu”, “Ideat”, “The Good Life”… The magazines of the I/O Media group resold to several buyers The A13 motorway closed in both directions for an “indefinite period” between Paris and Normandy
The A13 motorway closed in both directions for an “indefinite period” between Paris and Normandy The commitment to reduce taxes of 2 billion euros for households “will be kept”, assures Gabriel Attal
The commitment to reduce taxes of 2 billion euros for households “will be kept”, assures Gabriel Attal The exclusive Vespa that pays tribute to 140 years of Piaggio
The exclusive Vespa that pays tribute to 140 years of Piaggio Kingdom of the great maxi scooters: few and Kymco wants the crown of the Yamaha TMax
Kingdom of the great maxi scooters: few and Kymco wants the crown of the Yamaha TMax A complaint filed against Kanye West, accused of hitting an individual who had just attacked his wife
A complaint filed against Kanye West, accused of hitting an individual who had just attacked his wife In Béarn, a call for donations to renovate the house of Henri IV's mother
In Béarn, a call for donations to renovate the house of Henri IV's mother Skoda Kodiaq 2024: a 'beast' plug-in hybrid SUV
Skoda Kodiaq 2024: a 'beast' plug-in hybrid SUV Tesla launches a new Model Y with 600 km of autonomy at a "more accessible price"
Tesla launches a new Model Y with 600 km of autonomy at a "more accessible price" The 10 best-selling cars in March 2024 in Spain: sales fall due to Easter
The 10 best-selling cars in March 2024 in Spain: sales fall due to Easter A private jet company buys more than 100 flying cars
A private jet company buys more than 100 flying cars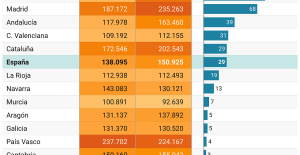 This is how housing prices have changed in Spain in the last decade
This is how housing prices have changed in Spain in the last decade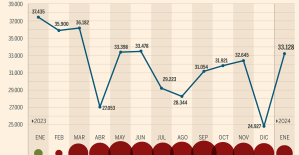 The home mortgage firm drops 10% in January and interest soars to 3.46%
The home mortgage firm drops 10% in January and interest soars to 3.46% The jewel of the Rocío de Nagüeles urbanization: a dream villa in Marbella
The jewel of the Rocío de Nagüeles urbanization: a dream villa in Marbella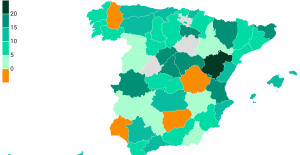 Rental prices grow by 7.3% in February: where does it go up and where does it go down?
Rental prices grow by 7.3% in February: where does it go up and where does it go down? With the promise of a “real burst of authority”, Gabriel Attal provokes the ire of the opposition
With the promise of a “real burst of authority”, Gabriel Attal provokes the ire of the opposition Europeans: the schedule of debates to follow between now and June 9
Europeans: the schedule of debates to follow between now and June 9 Europeans: “In France, there is a left and there is a right,” assures Bellamy
Europeans: “In France, there is a left and there is a right,” assures Bellamy During the night of the economy, the right points out the budgetary flaws of the macronie
During the night of the economy, the right points out the budgetary flaws of the macronie These French cities that will boycott the World Cup in Qatar
These French cities that will boycott the World Cup in Qatar Women's C1: at what time and on which channel to watch the OL-PSG semi-final first leg
Women's C1: at what time and on which channel to watch the OL-PSG semi-final first leg Tennis: after two victories this Friday, Grégoire Barrère qualifies for the semi-finals of the Bucharest tournament
Tennis: after two victories this Friday, Grégoire Barrère qualifies for the semi-finals of the Bucharest tournament Cycling: Mathieu Van der Poel “recharged the batteries” for Liège-Bastogne-Liège
Cycling: Mathieu Van der Poel “recharged the batteries” for Liège-Bastogne-Liège Mercato: Zidane at Bayern? We'll talk about it again, but...
Mercato: Zidane at Bayern? We'll talk about it again, but...


















