According to two sources, U.S. health officials are likely to recommend that all Americans receive an additional dose of the vaccine eight months after their second shot. This was according to two people who spoke with The Associated Press under anonymity to discuss internal discussions.
This means that the largest vaccination drive in American history is set to intensify.
This is due to both the highly contagious variant of the disease and preliminary evidence that vaccine protection begins to decrease within months.
This is why the U.S. has reacted with urgency to the situation. President Joe Biden declared independence from the virus on July Fourth. However, since then, the number of infections, deaths, and hospitalizations has risen across the country, overcrowding many emergency rooms in the South and West.
Officials in the United States retorted that Pfizer's July announcement of plans to apply for approval for boosters was premature. Experts feared that a new appeal for boosters could impede the ongoing campaign to win over the millions of Americans who remain skeptical or hesitant about getting their first shot.
"We need to make sure that, while we spend a lot on third doses, that we don’t undermine our campaign of first vaccinations," Lawrence Gostin (a Georgetown University public health specialist), said Tuesday. "That's the real existential crisis in America."
Gostin warned that calling for third doses may discourage people who were skeptical about the effectiveness of the shot.
Once the Food and Drug Administration officially approves the vaccines, booster shots will only be available to the general public. Currently, the vaccines are being distributed under an emergency use authorization. The Pfizer shot will be fully approved in the coming weeks.
U.S. officials recommended that boosters be given to some patients with weak immune systems last week, including cancer patients and transplant recipients.
Dr. Francis Collins, director of the National Institutes of Health said Sunday that the United States could decide within the next few weeks whether to provide booster shots to other Americans this fall.
They could be given to health care workers, residents of nursing homes, and older Americans who were among the first to get them.
According to the Centers for Disease Control and Prevention, more than 198 million Americans have had at least one dose (or 70%) of the COVID-19 vaccine. Only a little less than 60% of Americans aged 12 or older have been fully vaccinated.
Biden's administration was disappointed that the vaccination drive took place slower than they had expected. The variant is also spreading rapidly through unvaccinated areas, causing more "breakthrough infections" among fully-vaccinated people.
Although the vaccine is still highly effective against severe COVID-19 infection, studies have shown that its effectiveness has declined. However, Israel's latest results suggest that it may be less effective. Two months after the second dose, its effectiveness against symptomatic infections peaked at 96%. It was 90% four months later. It was at 84% after six months.
Israel, which only administered the Pfizer shot has offered a booster to those over 50 to stop its delta surge. Researchers are still trying understand if the breakthrough infections are caused by a decrease in immunity or vulnerability to the Delta variant.
European medical regulators stated Tuesday that they were in contact with vaccine developers regarding the need for boosters, but have not made any decisions.
Pfizer and BioNTech, its partner, announced Monday that they had submitted data to FDA in support of authorizing a booster shot. Pfizer stated that a small study found that people who received a third dose of coronavirus vaccine had higher levels than those who received the first. Pfizer is currently working on a larger study.
Americans who were given the first dose of Pfizer's vaccine as a health care worker or resident in a nursing home are now approaching eight months.
Collins, NIH's director of vaccines, stated that there is a risk that the vaccine might lose its effectiveness. We must deal with the problem of delta. Combining these two may mean we need boosters. This could be starting with health care providers and people in nursing homes. Then, gradually working with other people.
He stated that the variant had only begun to affect the country in July. The next few weeks will provide case data which will assist the U.S. in making a decision.
Two doses of the Moderna and Pfizer vaccines are given. Officials continue to gather information about the Johnson & Johnson vaccine that was approved in the U.S. late February. This will help determine when boosters should be recommended.
According to the White House, even though the U.S. is sharing more than 110 millions vaccine doses with other countries, it still has enough vaccines to give boosters to Americans.
The World Health Organization and other global health officials have asked richer countries to delay booster shots in order to supply first doses to people living in poverty.
Gostin stated that the Biden administration's plans for boosters to be recommended to all Americans is "a slap on the face" of other countries that have not been able to immunise their most vulnerable citizens.
He said, "It's tone deaf to all of the rest of the planet that can't even receive any doses."

 After 13 years of mission and seven successive leaders, the UN at an impasse in Libya
After 13 years of mission and seven successive leaders, the UN at an impasse in Libya Germany: search of AfD headquarters in Lower Saxony, amid accusations of embezzlement
Germany: search of AfD headquarters in Lower Saxony, amid accusations of embezzlement Faced with Iran, Israel plays appeasement and continues its shadow war
Faced with Iran, Israel plays appeasement and continues its shadow war Iran-Israel conflict: what we know about the events of the night after the explosions in Isfahan
Iran-Israel conflict: what we know about the events of the night after the explosions in Isfahan Sánchez condemns Iran's attack on Israel and calls for "containment" to avoid an escalation
Sánchez condemns Iran's attack on Israel and calls for "containment" to avoid an escalation China's GDP grows 5.3% in the first quarter, more than expected
China's GDP grows 5.3% in the first quarter, more than expected Alert on the return of whooping cough, a dangerous respiratory infection for babies
Alert on the return of whooping cough, a dangerous respiratory infection for babies Can relaxation, sophrology and meditation help with insomnia?
Can relaxation, sophrology and meditation help with insomnia? Vacation departures and returns: with the first crossovers, heavy traffic is expected this weekend
Vacation departures and returns: with the first crossovers, heavy traffic is expected this weekend “Têtu”, “Ideat”, “The Good Life”… The magazines of the I/O Media group resold to several buyers
“Têtu”, “Ideat”, “The Good Life”… The magazines of the I/O Media group resold to several buyers The A13 motorway closed in both directions for an “indefinite period” between Paris and Normandy
The A13 motorway closed in both directions for an “indefinite period” between Paris and Normandy The commitment to reduce taxes of 2 billion euros for households “will be kept”, assures Gabriel Attal
The commitment to reduce taxes of 2 billion euros for households “will be kept”, assures Gabriel Attal The exclusive Vespa that pays tribute to 140 years of Piaggio
The exclusive Vespa that pays tribute to 140 years of Piaggio Kingdom of the great maxi scooters: few and Kymco wants the crown of the Yamaha TMax
Kingdom of the great maxi scooters: few and Kymco wants the crown of the Yamaha TMax A complaint filed against Kanye West, accused of hitting an individual who had just attacked his wife
A complaint filed against Kanye West, accused of hitting an individual who had just attacked his wife In Béarn, a call for donations to renovate the house of Henri IV's mother
In Béarn, a call for donations to renovate the house of Henri IV's mother Skoda Kodiaq 2024: a 'beast' plug-in hybrid SUV
Skoda Kodiaq 2024: a 'beast' plug-in hybrid SUV Tesla launches a new Model Y with 600 km of autonomy at a "more accessible price"
Tesla launches a new Model Y with 600 km of autonomy at a "more accessible price" The 10 best-selling cars in March 2024 in Spain: sales fall due to Easter
The 10 best-selling cars in March 2024 in Spain: sales fall due to Easter A private jet company buys more than 100 flying cars
A private jet company buys more than 100 flying cars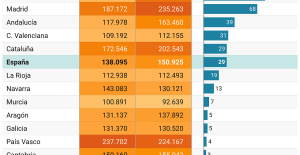 This is how housing prices have changed in Spain in the last decade
This is how housing prices have changed in Spain in the last decade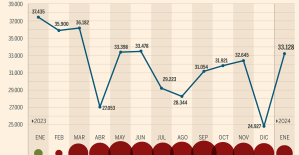 The home mortgage firm drops 10% in January and interest soars to 3.46%
The home mortgage firm drops 10% in January and interest soars to 3.46% The jewel of the Rocío de Nagüeles urbanization: a dream villa in Marbella
The jewel of the Rocío de Nagüeles urbanization: a dream villa in Marbella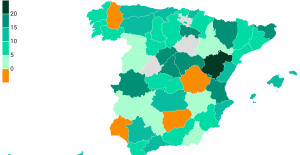 Rental prices grow by 7.3% in February: where does it go up and where does it go down?
Rental prices grow by 7.3% in February: where does it go up and where does it go down? With the promise of a “real burst of authority”, Gabriel Attal provokes the ire of the opposition
With the promise of a “real burst of authority”, Gabriel Attal provokes the ire of the opposition Europeans: the schedule of debates to follow between now and June 9
Europeans: the schedule of debates to follow between now and June 9 Europeans: “In France, there is a left and there is a right,” assures Bellamy
Europeans: “In France, there is a left and there is a right,” assures Bellamy During the night of the economy, the right points out the budgetary flaws of the macronie
During the night of the economy, the right points out the budgetary flaws of the macronie These French cities that will boycott the World Cup in Qatar
These French cities that will boycott the World Cup in Qatar Rally: Neuville and Evans neck and neck after the first day in Croatia
Rally: Neuville and Evans neck and neck after the first day in Croatia Gymnastics: after Rio and Tokyo, Frenchman Samir Aït Saïd qualified for the Paris 2024 Olympics
Gymnastics: after Rio and Tokyo, Frenchman Samir Aït Saïd qualified for the Paris 2024 Olympics Top 14: in the fight for maintenance, Perpignan has the wind at its back
Top 14: in the fight for maintenance, Perpignan has the wind at its back Top 14: Toulon-Toulouse, a necessarily special reunion for Melvyn Jaminet
Top 14: Toulon-Toulouse, a necessarily special reunion for Melvyn Jaminet


















