The new gene therapy Zolgensma not come out of the headlines. Because for the second Time Novartis have to admit that there have been studies to agents for the treatment of genetically determined muscular dystrophy (spinal muscular atrophy, a short-SMA) to a delayed message.
Zolgensma was developed by US Start-up Avexis, which Novartis had acquired last year for $ 8.7 billion. Gene therapy is approved in the United States, with a list price of $ 2.1 million Zolgensma is the most expensive drug in the world.
problems with animal
try Currently Zolgensma for the treatment of babies with the most severe Form of muscular disease, SMA type 1, is approved. Now, Avexis is working, the admission of the less serious forms of the disease to extend.
This Wednesday, the U.S. FDA has imposed a temporary stop for clinical studies with Zolgensma. In these studies children were treated up to the age of 5 years with the gene therapy, which are affected by the weaker variant of the disease, SMA type 2. The trigger for the study stop results from animal experiments in the preclinical Phase.
This monkey Zolgensma was injected via a Spinal injection. In Tests, the highest dose with the medium-heavy side effects, such as neuronal cellular inflammation, which can lead to cell loss.
problems since March known
The problems however were not new. Avexis knew of the side effects of the use of animal testing since March. A report to the industry portal "Fierce Pharma" confirmed Novartis.
Therefore, for the responsible Manager of Avexis learned in March of the problems. It was decided to report the results in the annual Update of the authorities, which was scheduled for September. "Unfortunately, an error was made, and the Update has not been implemented," said Novartis. The FDA was not informed until the end of last week.
The Occurrence of side effects in trials with monkeys he had noticed, as Novartis of the data manipulation was engaged, which had taken care of this summer, headlines around the world.
Tests had no relevance at all
researchers from Avexis had manipulated in the development of gene therapy results of Tests with mice. Thanks to an internal Tipster Novartis already knew since 14. March of the case. However, the group announced that the U.S. food and drug administration on may 28. June – good month, after the FDA had granted approval for Zolgensma for the treatment of SMA type 1. The U.S. food and drug administration was criticized by media release this late message is sharp, and the case made public.
Novartis had substantiated at the time the late Information that the group wanted to wait for the results of a two-piece internal investigation of the case. Among other things, the question of the mice tests were carried out again to examine whether any Changes to the application for authorisation were necessary. In fact, the Tests had no relevance to the efficacy, safety or tolerability of Zolgensma.
FDA responded within a matter of days
The processing of that data manipulation is supported now, so the delay in reporting the problems at the animal to light try. It is striking that the U.S. FDA has now responded within a few days. And the temporary test ban imposed.
A spokesman for Novartis, said that the group pursuing the question of why the message about the problems with animal late was made and what consequences to draw from it.
the group is a lot on the game. The current approval of Zolgensma is not in doubt. The funds will continue to be sold. However, the test ban could be delayed, that the FDA allows gene therapy for other fields of application. The analysts of Vontobel appreciate that the admission of Zolgensma for type 2 the muscle weakness in SMA could bring in an additional revenue of up to $ 800 million per year.
Created: 01.11.2019, 15:26 PM

 Germany: Man armed with machete enters university library and threatens staff
Germany: Man armed with machete enters university library and threatens staff His body naturally produces alcohol, he is acquitted after a drunk driving conviction
His body naturally produces alcohol, he is acquitted after a drunk driving conviction Who is David Pecker, the first key witness in Donald Trump's trial?
Who is David Pecker, the first key witness in Donald Trump's trial? What does the law on the expulsion of migrants to Rwanda adopted by the British Parliament contain?
What does the law on the expulsion of migrants to Rwanda adopted by the British Parliament contain? Parvovirus alert, the “fifth disease” of children which has already caused the death of five babies in 2024
Parvovirus alert, the “fifth disease” of children which has already caused the death of five babies in 2024 Colorectal cancer: what to watch out for in those under 50
Colorectal cancer: what to watch out for in those under 50 H5N1 virus: traces detected in pasteurized milk in the United States
H5N1 virus: traces detected in pasteurized milk in the United States What High Blood Pressure Does to Your Body (And Why It Should Be Treated)
What High Blood Pressure Does to Your Body (And Why It Should Be Treated) Insurance: SFAM, subsidiary of Indexia, placed in compulsory liquidation
Insurance: SFAM, subsidiary of Indexia, placed in compulsory liquidation Under pressure from Brussels, TikTok deactivates the controversial mechanisms of its TikTok Lite application
Under pressure from Brussels, TikTok deactivates the controversial mechanisms of its TikTok Lite application “I can’t help but panic”: these passengers worried about incidents on Boeing
“I can’t help but panic”: these passengers worried about incidents on Boeing “I’m interested in knowing where the money that the State takes from me goes”: Bruno Le Maire’s strange pay slip sparks controversy
“I’m interested in knowing where the money that the State takes from me goes”: Bruno Le Maire’s strange pay slip sparks controversy 25 years later, the actors of Blair Witch Project are still demanding money to match the film's record profits
25 years later, the actors of Blair Witch Project are still demanding money to match the film's record profits At La Scala, Mathilde Charbonneaux is Madame M., Jacqueline Maillan
At La Scala, Mathilde Charbonneaux is Madame M., Jacqueline Maillan Deprived of Hollywood and Western music, Russia gives in to the charms of K-pop and manga
Deprived of Hollywood and Western music, Russia gives in to the charms of K-pop and manga Exhibition: Toni Grand, the incredible odyssey of a sculptural thinker
Exhibition: Toni Grand, the incredible odyssey of a sculptural thinker Skoda Kodiaq 2024: a 'beast' plug-in hybrid SUV
Skoda Kodiaq 2024: a 'beast' plug-in hybrid SUV Tesla launches a new Model Y with 600 km of autonomy at a "more accessible price"
Tesla launches a new Model Y with 600 km of autonomy at a "more accessible price" The 10 best-selling cars in March 2024 in Spain: sales fall due to Easter
The 10 best-selling cars in March 2024 in Spain: sales fall due to Easter A private jet company buys more than 100 flying cars
A private jet company buys more than 100 flying cars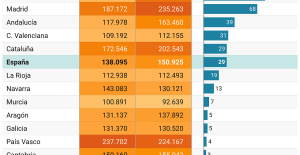 This is how housing prices have changed in Spain in the last decade
This is how housing prices have changed in Spain in the last decade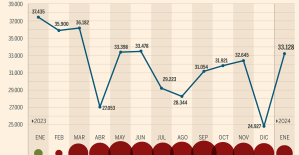 The home mortgage firm drops 10% in January and interest soars to 3.46%
The home mortgage firm drops 10% in January and interest soars to 3.46% The jewel of the Rocío de Nagüeles urbanization: a dream villa in Marbella
The jewel of the Rocío de Nagüeles urbanization: a dream villa in Marbella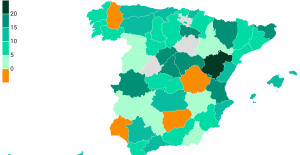 Rental prices grow by 7.3% in February: where does it go up and where does it go down?
Rental prices grow by 7.3% in February: where does it go up and where does it go down? Sale of Biogaran: The Republicans write to Emmanuel Macron
Sale of Biogaran: The Republicans write to Emmanuel Macron Europeans: “All those who claim that we don’t need Europe are liars”, criticizes Bayrou
Europeans: “All those who claim that we don’t need Europe are liars”, criticizes Bayrou With the promise of a “real burst of authority”, Gabriel Attal provokes the ire of the opposition
With the promise of a “real burst of authority”, Gabriel Attal provokes the ire of the opposition Europeans: the schedule of debates to follow between now and June 9
Europeans: the schedule of debates to follow between now and June 9 These French cities that will boycott the World Cup in Qatar
These French cities that will boycott the World Cup in Qatar Hand: Montpellier crushes Kiel and continues to dream of the Champions League
Hand: Montpellier crushes Kiel and continues to dream of the Champions League OM-Nice: a spectacular derby, Niçois timid despite their numerical superiority...The tops and the flops
OM-Nice: a spectacular derby, Niçois timid despite their numerical superiority...The tops and the flops Tennis: 1000 matches and 10 notable encounters by Richard Gasquet
Tennis: 1000 matches and 10 notable encounters by Richard Gasquet Tennis: first victory of the season on clay for Osaka in Madrid
Tennis: first victory of the season on clay for Osaka in Madrid


















