Packaging, shampoos, non-stick pans or even make-up... Perfluorinated chemicals, or PFAS, adorn the ingredient list of countless consumer products. The industry loves these molecule families. However, according to a study published last week, all of the rainwater on Earth is unfit for consumption, in particular because of the presence of these compounds which are toxic for living organisms and for humans.
Until now, PFAS were nicknamed the "eternal pollutants". Due to their composition, they persist almost indefinitely in nature and in living organisms. But since Thursday, August 18, the invincibility of the PFAS no longer seems so certain. Scientists have announced that they have found a relatively affordable and simple method to destroy these compounds which, above a certain dose, can cause cancer, fetal malformations, infertility or an increase in cholesterol levels, among other things.
The technique discovered requires relatively low temperatures and reagents common in industry. It was developed by chemists in the United States and China whose work has been published in the scientific journal Science. A surprise for the researchers behind this success: "It's exciting to see how simple and yet unknown our solution is", recognizes William Dichtel, researcher in chemistry for the University of Northwestern William and co-author of the 'study.
The scientists started from an observation: "PFAS have become a major social problem", continues William Dichtel in a press release. For decades, some American companies have dumped these products into rivers, contaminating many drinking waters and having to launch huge clean-up companies. In the United States, 97% of the American population has perfluorinated products in their blood, reports Northwestern University.
In France, recent discharges have been discovered near Lyon and are the subject of an investigation. But through their resistance, the PFAS are spreading outside of major scandals. According to a study published in 2019 by Public Health France, out of the thousand individuals observed, none had an organism spared by these molecules. But "even a very small amount of certain PFAS has negative effects on health", recalls William Dichtel. Many water filters have been set up to separate these products. But what to do next?
Until now, methods for getting rid of these pollutants have been particularly cumbersome, requiring very high temperature incineration or ultrasonic irradiation. Largely insufficient. "We can't just wait for this problem to be solved. We wanted to use chemistry to tackle this problem and create a solution that the world can use", explains the researcher. The resistance of PFAS results from the bonds between the carbon and fluorine atoms that compose them. These bonds are among the strongest known in organic chemistry.
Results: some methods used so far have proven to be counterproductive. "In New York State, we found that a plant claiming to incinerate PFAS was actually releasing some of these compounds into the air," Dichtel said. “Others have buried the compounds in landfills. This is the promise of seeing the problem reappear in 30 years, as PFAS are gradually spreading in the ground,” he added.
But the "eternal pollutants" have a weak point. At one end of their molecule, a cluster of oxygen atoms can be targeted by a solvent and reagent common in industry, at average temperatures of 80 to 120 degrees Celsius. "This causes the entire molecule to collapse in a cascade of complex reactions," says William Dichtel. The scientists then mapped all of these reactions, and the mechanisms triggering them. What better to know this method of destruction and potentially improve it.
While this discovery is important, it does not solve the PFAS problem. Because this technique works on only ten PFAS. However, 12,000 have been identified in the wild by the US Environmental Protection Agency. "There are other types of PFAS that don't have the same Achilles heel, but each has its own weak point," said William Dichtel, who promises to continue his efforts. "If we can identify it, then we will know how to activate it to destroy it."
If the method acts on the molecule, it is still necessary to succeed in filtering the latter from the waters and the organisms. At the same time, national institutions have therefore begun to regulate their use and monitor releases. The European Union plans, for its part, to eliminate them from consumer products by 2030. Do not pollute, rather than depollute.

 Germany: Man armed with machete enters university library and threatens staff
Germany: Man armed with machete enters university library and threatens staff His body naturally produces alcohol, he is acquitted after a drunk driving conviction
His body naturally produces alcohol, he is acquitted after a drunk driving conviction Who is David Pecker, the first key witness in Donald Trump's trial?
Who is David Pecker, the first key witness in Donald Trump's trial? What does the law on the expulsion of migrants to Rwanda adopted by the British Parliament contain?
What does the law on the expulsion of migrants to Rwanda adopted by the British Parliament contain?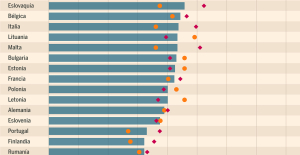 Spain is the country in the European Union with the most overqualified workers for their jobs
Spain is the country in the European Union with the most overqualified workers for their jobs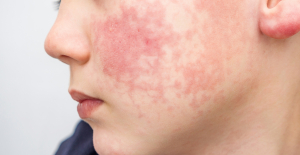 Parvovirus alert, the “fifth disease” of children which has already caused the death of five babies in 2024
Parvovirus alert, the “fifth disease” of children which has already caused the death of five babies in 2024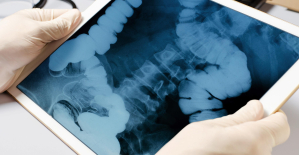 Colorectal cancer: what to watch out for in those under 50
Colorectal cancer: what to watch out for in those under 50 H5N1 virus: traces detected in pasteurized milk in the United States
H5N1 virus: traces detected in pasteurized milk in the United States Insurance: SFAM, subsidiary of Indexia, placed in compulsory liquidation
Insurance: SFAM, subsidiary of Indexia, placed in compulsory liquidation Under pressure from Brussels, TikTok deactivates the controversial mechanisms of its TikTok Lite application
Under pressure from Brussels, TikTok deactivates the controversial mechanisms of its TikTok Lite application “I can’t help but panic”: these passengers worried about incidents on Boeing
“I can’t help but panic”: these passengers worried about incidents on Boeing “I’m interested in knowing where the money that the State takes from me goes”: Bruno Le Maire’s strange pay slip sparks controversy
“I’m interested in knowing where the money that the State takes from me goes”: Bruno Le Maire’s strange pay slip sparks controversy 25 years later, the actors of Blair Witch Project are still demanding money to match the film's record profits
25 years later, the actors of Blair Witch Project are still demanding money to match the film's record profits At La Scala, Mathilde Charbonneaux is Madame M., Jacqueline Maillan
At La Scala, Mathilde Charbonneaux is Madame M., Jacqueline Maillan Deprived of Hollywood and Western music, Russia gives in to the charms of K-pop and manga
Deprived of Hollywood and Western music, Russia gives in to the charms of K-pop and manga Exhibition: Toni Grand, the incredible odyssey of a sculptural thinker
Exhibition: Toni Grand, the incredible odyssey of a sculptural thinker Skoda Kodiaq 2024: a 'beast' plug-in hybrid SUV
Skoda Kodiaq 2024: a 'beast' plug-in hybrid SUV Tesla launches a new Model Y with 600 km of autonomy at a "more accessible price"
Tesla launches a new Model Y with 600 km of autonomy at a "more accessible price" The 10 best-selling cars in March 2024 in Spain: sales fall due to Easter
The 10 best-selling cars in March 2024 in Spain: sales fall due to Easter A private jet company buys more than 100 flying cars
A private jet company buys more than 100 flying cars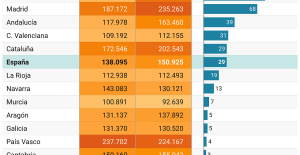 This is how housing prices have changed in Spain in the last decade
This is how housing prices have changed in Spain in the last decade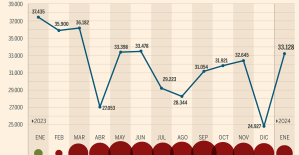 The home mortgage firm drops 10% in January and interest soars to 3.46%
The home mortgage firm drops 10% in January and interest soars to 3.46% The jewel of the Rocío de Nagüeles urbanization: a dream villa in Marbella
The jewel of the Rocío de Nagüeles urbanization: a dream villa in Marbella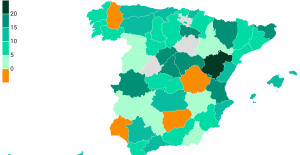 Rental prices grow by 7.3% in February: where does it go up and where does it go down?
Rental prices grow by 7.3% in February: where does it go up and where does it go down? Sale of Biogaran: The Republicans write to Emmanuel Macron
Sale of Biogaran: The Republicans write to Emmanuel Macron Europeans: “All those who claim that we don’t need Europe are liars”, criticizes Bayrou
Europeans: “All those who claim that we don’t need Europe are liars”, criticizes Bayrou With the promise of a “real burst of authority”, Gabriel Attal provokes the ire of the opposition
With the promise of a “real burst of authority”, Gabriel Attal provokes the ire of the opposition Europeans: the schedule of debates to follow between now and June 9
Europeans: the schedule of debates to follow between now and June 9 These French cities that will boycott the World Cup in Qatar
These French cities that will boycott the World Cup in Qatar Hand: Montpellier crushes Kiel and continues to dream of the Champions League
Hand: Montpellier crushes Kiel and continues to dream of the Champions League OM-Nice: a spectacular derby, Niçois timid despite their numerical superiority...The tops and the flops
OM-Nice: a spectacular derby, Niçois timid despite their numerical superiority...The tops and the flops Tennis: 1000 matches and 10 notable encounters by Richard Gasquet
Tennis: 1000 matches and 10 notable encounters by Richard Gasquet Tennis: first victory of the season on clay for Osaka in Madrid
Tennis: first victory of the season on clay for Osaka in Madrid


















