the world health organisation, WHO, says, that it can only approve a Covid-19-vaccine, if it has been through strict safety tests. The opinion is a comment to Russia's surprising announcement that it has approved a vaccine against the new coronavirus.
on Tuesday morning, for the first time in the world registered a vaccine in Russia against the new coronavirus, said president Vladimir Putin in a televised video calls to his ministers on Tuesday morning.
In Geneva, says WHO spokesman Tarik Seward, that the united nations organization is in close contact with the Russian health authorities, and to hold discussions about WHO's view of the vaccine.
Russia's Sputnik V vaccine has been developed by the research institute Gamaleja in cooperation with the Russian ministry of defense.
There is work currently on the 165 candidate vaccines all over the world, according to the WHO's latest overview. 26 of the vaccines is being tested on humans. Six vaccines are most developed and in the so-called Phase 3 clinical evaluation.
Gamaleja-the candidate is among the 26 which are tested on people.
It is detected as being in phase 1. A Russian spokesman says that the Wednesday will be moved to phase 3, and that industrial production will begin in september. Three western vaccines and two chinese are in Phase 3 now. 20 countries have ordered over a billion doses, it says.
In the world as a whole, countries have ordered 5.7 billion doses of the different candidate vaccines, when they become ready.
Oxford University, which has collaborated with medicinalgiganten AstraZeneca, hope on the results in september.
The White House says president Trumps special adviser Kellyanne Conway, that Russia has not undertaken the necessary investigations.
A spokeswoman for the German ministry of health says that there are doubts about the quality of the new vaccine from Russia.
- the EU approves only of new vaccines and new medicines, which are fully clinically tested, she says.

 Sudan ravaged by a year of war
Sudan ravaged by a year of war Ten years after the kidnapping of the Chibok girls, what has become of the terrorist group Boko Haram?
Ten years after the kidnapping of the Chibok girls, what has become of the terrorist group Boko Haram? The Israeli government divided on the extent of its response after Iran's direct attack on its territory
The Israeli government divided on the extent of its response after Iran's direct attack on its territory “Is it time for a killing blow?” : the Israeli press wonders about a response after the Iranian attack
“Is it time for a killing blow?” : the Israeli press wonders about a response after the Iranian attack Covid-19: everything you need to know about the new vaccination campaign which is starting
Covid-19: everything you need to know about the new vaccination campaign which is starting The best laptops of the moment boast artificial intelligence
The best laptops of the moment boast artificial intelligence Amazon invests 700 million in robotizing its warehouses in Europe
Amazon invests 700 million in robotizing its warehouses in Europe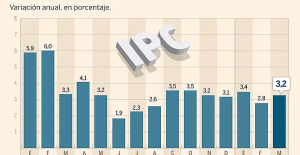 Inflation rises to 3.2% in March due to gasoline and electricity bills
Inflation rises to 3.2% in March due to gasoline and electricity bills Large French companies are alarmed by the stalling of the European economy
Large French companies are alarmed by the stalling of the European economy New sanctions against Russia send industrial metal prices soaring
New sanctions against Russia send industrial metal prices soaring Olympic Games 2024: civil servants authorized to carry out private security activities
Olympic Games 2024: civil servants authorized to carry out private security activities Threat of strikes for the 2024 Olympics: Macron says he has “confidence” in the “spirit of responsibility” of the unions
Threat of strikes for the 2024 Olympics: Macron says he has “confidence” in the “spirit of responsibility” of the unions Musical show: “Come Bach”, fantasies around the composer
Musical show: “Come Bach”, fantasies around the composer The cry of alarm from Senghor's friends has been heard
The cry of alarm from Senghor's friends has been heard World Chess Championship: Nepomniachtchi and Gukesh lead the candidates tournament
World Chess Championship: Nepomniachtchi and Gukesh lead the candidates tournament Rapper Heuss L’Enfoiré calls on Ronaldinho for his new music video
Rapper Heuss L’Enfoiré calls on Ronaldinho for his new music video Skoda Kodiaq 2024: a 'beast' plug-in hybrid SUV
Skoda Kodiaq 2024: a 'beast' plug-in hybrid SUV Tesla launches a new Model Y with 600 km of autonomy at a "more accessible price"
Tesla launches a new Model Y with 600 km of autonomy at a "more accessible price" The 10 best-selling cars in March 2024 in Spain: sales fall due to Easter
The 10 best-selling cars in March 2024 in Spain: sales fall due to Easter A private jet company buys more than 100 flying cars
A private jet company buys more than 100 flying cars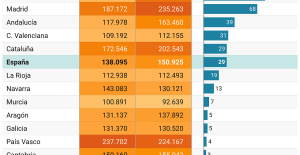 This is how housing prices have changed in Spain in the last decade
This is how housing prices have changed in Spain in the last decade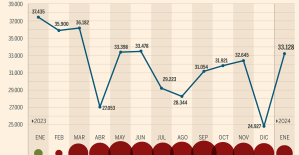 The home mortgage firm drops 10% in January and interest soars to 3.46%
The home mortgage firm drops 10% in January and interest soars to 3.46% The jewel of the Rocío de Nagüeles urbanization: a dream villa in Marbella
The jewel of the Rocío de Nagüeles urbanization: a dream villa in Marbella Rental prices grow by 7.3% in February: where does it go up and where does it go down?
Rental prices grow by 7.3% in February: where does it go up and where does it go down? Europeans: the schedule of debates to follow between now and June 9
Europeans: the schedule of debates to follow between now and June 9 Europeans: “In France, there is a left and there is a right,” assures Bellamy
Europeans: “In France, there is a left and there is a right,” assures Bellamy During the night of the economy, the right points out the budgetary flaws of the macronie
During the night of the economy, the right points out the budgetary flaws of the macronie Europeans: Glucksmann denounces “Emmanuel Macron’s failure” in the face of Bardella’s success
Europeans: Glucksmann denounces “Emmanuel Macron’s failure” in the face of Bardella’s success These French cities that will boycott the World Cup in Qatar
These French cities that will boycott the World Cup in Qatar Augusta Masters: the unchallenged reign of Scottie Scheffler
Augusta Masters: the unchallenged reign of Scottie Scheffler Tennis: Harold Mayot takes Cachin and goes to the second round in Barcelona
Tennis: Harold Mayot takes Cachin and goes to the second round in Barcelona Barça-PSG: “We will do it”, Luis Enrique shows his confidence before the return match
Barça-PSG: “We will do it”, Luis Enrique shows his confidence before the return match Hakimi on Mbappé before Barça-PSG: “I feel very good, very motivated, like the whole team”
Hakimi on Mbappé before Barça-PSG: “I feel very good, very motivated, like the whole team”


















